RNA Therapeutics for Cardiovascular Disease
Introduction
RNA therapeutics have recently gained global attention following their pivotal role in the first vaccines developed. This represents only one example of the broader therapeutic potential of RNA. RNA functions as a versatile biological software that can be rapidly designed and produced, enabling precise modulation of the expression or activity of virtually any clinically relevant protein. RNA-based therapeutics offer diverse applications, including vaccines for infectious diseases and cancer, enzyme replacement therapies, and enhancements in cell-based therapies, with additional potential in areas such as cardiovascular diseases. Recent advances in RNA synthesis, sequence design, and delivery technologies have made it feasible to translate RNA biology into clinically viable therapies.
RNA Mechanisms of Action
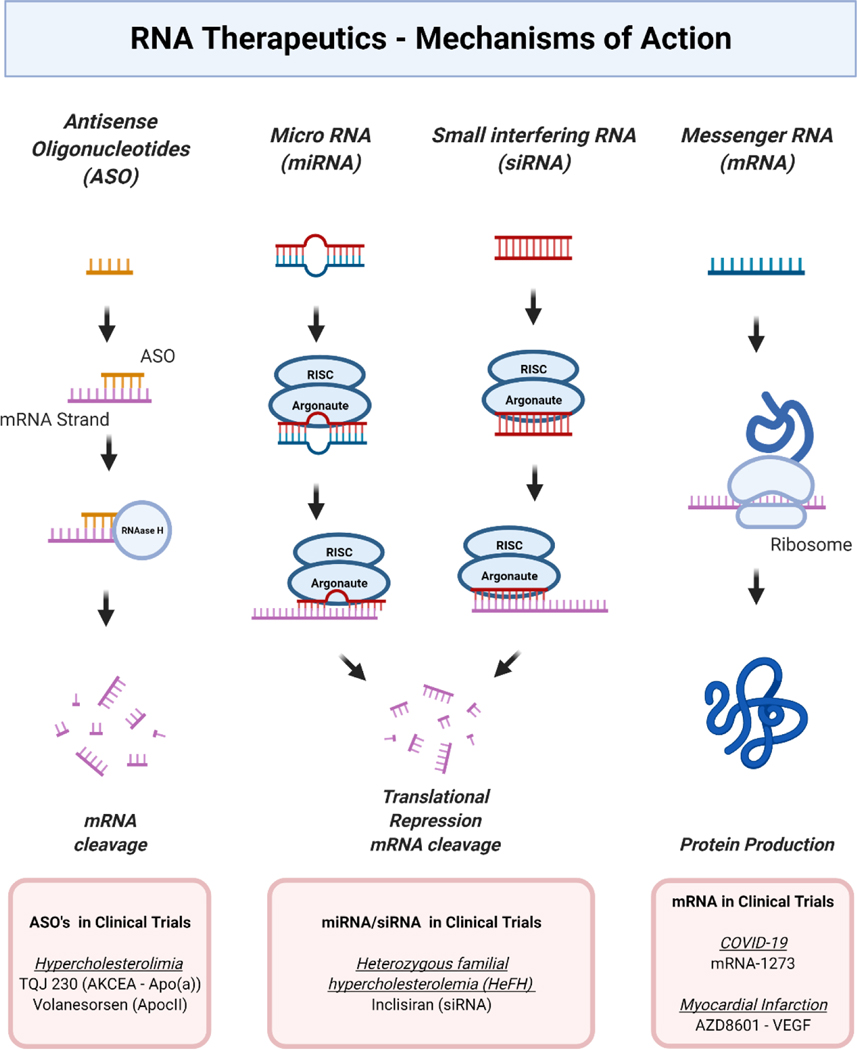
Messenger RNAs (mRNAs) serve as transient blueprints of genes encoded in genomic DNA and are translated by the cellular machinery to produce the corresponding proteins. RNA-based therapeutics can be designed either to introduce exogenous mRNA into cells or to inhibit the translation of endogenous mRNA.
Silencing endogenous mRNA can be achieved using antisense oligonucleotides (ASOs), small interfering RNAs (siRNAs), or microRNAs (miRNAs). While all bind complementary sequences on target mRNAs to prevent translation, their molecular mechanisms differ. ASOs are short single-stranded nucleic acids that form duplexes with target mRNA, recruiting RNase H to degrade the transcript or sterically blocking mRNA processing and translation. In contrast, siRNAs and miRNAs are non-coding duplexes processed by cellular machinery and incorporated into the RNA-induced silencing complex (RISC), which either cleaves the target mRNA or represses its translation. siRNAs are fully complementary and highly specific to a single gene, whereas miRNAs are partially complementary and can regulate multiple genes. Second-generation small RNAs, including agomirs and antagomirs, are chemically stabilized constructs that enhance in vivo efficacy. Agomirs mimic mature miRNAs to silence target transcripts, while antagomirs constitutively inhibit endogenous miRNAs. Chemical modifications such as 2′-O-methylation, phosphorothioate linkages, and cholesterol conjugation improve both RNA stability and cellular uptake.
Exogenous mRNA therapeutics supply the cell with a transcript encoding a therapeutic protein, enabling protein replacement, antigen expression for vaccination, or chimeric antigen receptor production for cell therapy. Unlike silencing RNAs, mRNA constructs are typically thousands of nucleotides long, allowing the expression of virtually any protein, including previously “undruggable” targets. Translation occurs in the cytoplasm without nuclear entry, reducing the risk of genomic integration associated with DNA-based therapies. mRNA sequences can also be modified for personalized therapeutics.
The first demonstration of synthetic mRNA efficacy occurred in 1992, when mRNA encoding vasopressin injected into the hypothalamus of rats with central diabetes insipidus transiently reversed the disease. Although RNA therapeutics can be manufactured via chemical synthesis or in vitro transcription, early applications were limited by activation of innate immune responses, causing inflammation and apoptosis. These challenges were overcome through nucleotide chemical modifications, purification methods to remove immunogenic byproducts, sequence optimization of UTRs and coding regions, mRNA circularization to enhance stability, and lyophilization to extend shelf life. Collectively, these advances have made mRNA therapeutics clinically viable, although efficient delivery remains a critical challenge, particularly for cardiovascular applications.
miRNA and siRNA utilize the RNA-induced silencing complex (RISC) to degrade complementary mRNA strands or block translation, thereby downregulating expression of specific target genes. Antisense oligonucleotides (ASOs) employ mechanisms such as steric hindrance or RNase H-mediated decay of pre-mRNA to inhibit translation. In contrast, therapeutic mRNA engages the cytoplasmic translational machinery to produce proteins that are beneficial for disease treatment. Silencing endogenous mRNA has been applied successfully in hypercholesterolemia, while expression of target proteins has been effective in vaccines for emily and shows potential in therapies for myocardial infarction.
Cardiovascular Delivery of RNA Therapeutics
RNA therapeutics have emerged as a versatile platform for modulating protein expression, offering applications in vaccines, protein replacement, gene editing, and cell therapy. mRNA therapeutics deliver exogenous transcripts to the cytoplasm for protein expression, while silencing RNAs such as siRNA, miRNA, and ASOs downregulate endogenous gene expression via RISC-mediated cleavage or steric and RNase H mechanisms. Advances in chemical modifications, sequence optimization, and purification have improved RNA stability, reduced immunogenicity, and enhanced translational efficiency.
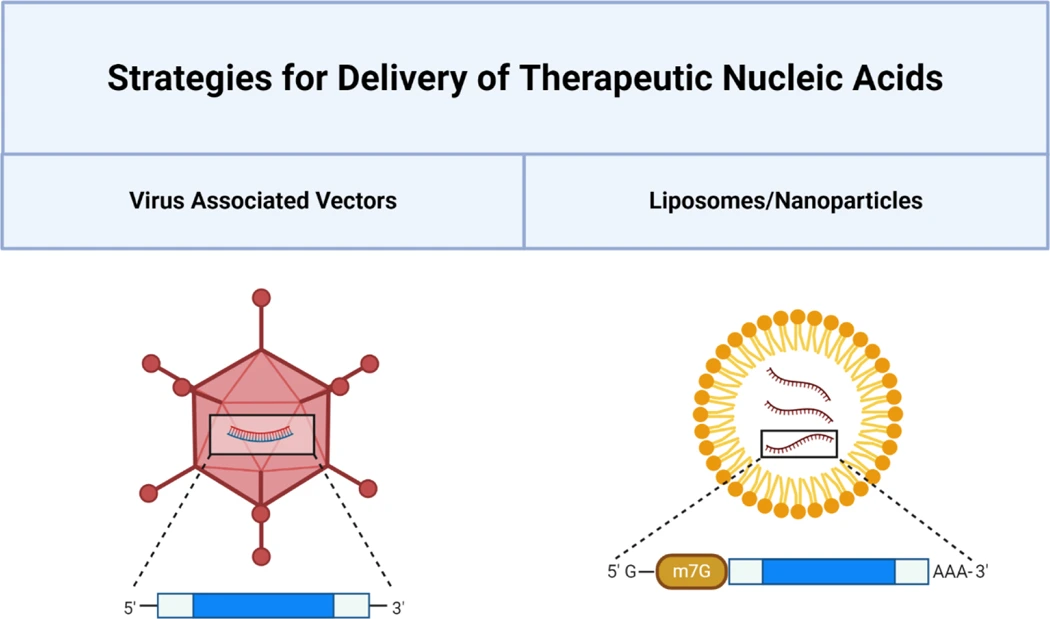
Efficient intracellular delivery remains a key challenge due to RNA’s large size, negative charge, and susceptibility to enzymatic degradation. Formulation strategies including lipid and polymer nanoparticles, lipoplexes, and leukocyte-mimetic biomimetic nanoparticles protect RNA, facilitate cellular uptake, and enable tissue-specific targeting. Molecular engineering approaches, such as 5’ cap modifications, poly(A) tail elongation, UTR and ORF optimization, and incorporation of modified nucleotides, further enhance mRNA half-life and protein expression. Cell-specific targeting can increase efficacy while reducing off-target effects, exemplified by dendritic cell-targeted nanoparticles and leukosome platforms for cardiovascular disease.
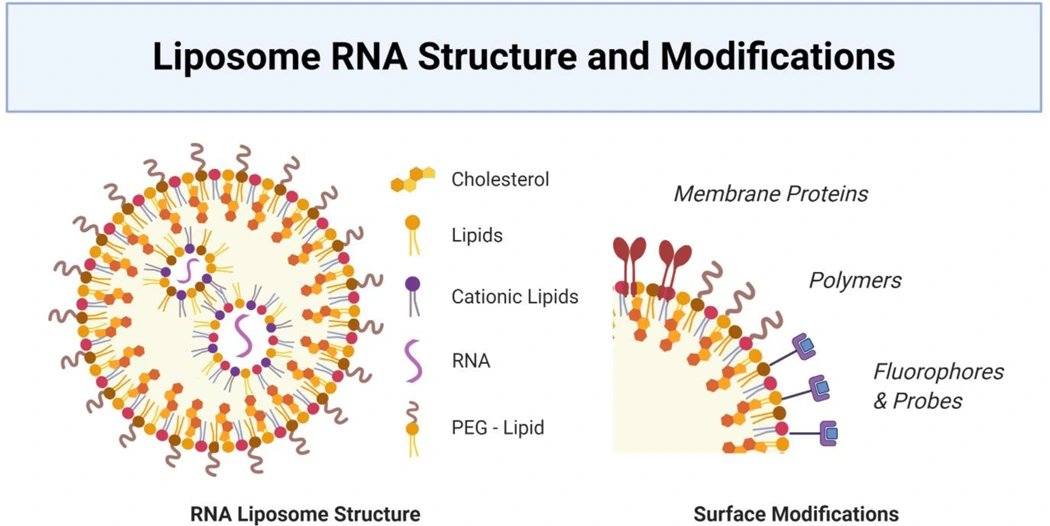
In cardiovascular applications, RNA delivery exploits both local and systemic targeting, leveraging hemodynamic differences, activated endothelium, or organ specific uptake ( liver or bone marrow). Preclinical studies demonstrate the potential for RNA therapeutics to modulate endothelial function, enhance cardiac repair post-infarction, and stabilize atherosclerotic plaques.
Collectively, RNA therapeutics provide a flexible and rapidly deployable platform capable of addressing previously undruggable targets, with ongoing advances in stability, delivery, and targeting expanding their clinical potential. Future developments will likely focus on precise tissue-specific delivery, optimized nanoparticle platforms, and continued integration of chemical and structural RNA modifications to maximize safety, efficacy, and translational feasibility.
RNA Therapeutics Currently in Clinical Trials
Cardiovascular disease remains the leading cause of mortality worldwide, accounting for approximately 17.8 million deaths annually. While clinical studies of RNA therapeutics for cardiovascular indications are still limited, ongoing research and investment suggest a growing pipeline of candidates.
One of the earliest nucleic acid therapies for cardiovascular disease was Mipomersen, a second-generation phosphorothioate antisense oligonucleotide (ASO) targeting apolipoprotein B-100 mRNA. Initially approved for homozygous familial hypercholesterolemia, Mipomersen reduced LDL cholesterol without elevating C-reactive protein but was later discontinued due to severe liver toxicity. Subsequent ASOs have been chemically optimized to reduce adverse effects. For example, TQJ230 (AKCEA-APO(a)-LRx), an ASO targeting lipoprotein(a) mRNA, achieved lipoprotein(a) levels below 50 mg/dL in over 90% of patients in a phase II study. Volanesorsen, another ASO, binds APOC3 mRNA and promotes its degradation, resulting in reductions in Apoc3 (−80%) and triglycerides (−71%) while increasing HDL-C (46%). These examples demonstrate that chemical modifications have significantly improved the safety and efficacy of ASO-based cardiovascular therapies.
Another strategy targets PCSK9 mRNA in the liver, which normally promotes LDL receptor degradation and increases plasma LDL cholesterol. Inclisiran, a chemically modified siRNA conjugated to a triantennary N-acetylgalactosamine (GalNAc) ligand, has shown comparable LDL-lowering efficacy to monoclonal antibody PCSK9 inhibitors (alirocumab, evolocumab) with twice-yearly subcutaneous injections. Inclisiran reduces LDL cholesterol by 40–50% in patients with severe hypercholesterolemia and has been approved by the European Commission for treatment-resistant cases.
Beyond gene-silencing approaches, VEGF-A mRNA (Moderna) represents the first cardiovascular-related mRNA therapy in clinical trials. In a phase I study, intradermal VEGF-A mRNA increased local protein expression and improved skin perfusion in patients with type 2 diabetes. The ongoing phase 2a EPICCURE trial will assess whether epicardial injections of VEGF-A mRNA can restore ischemic but viable myocardial tissue in patients undergoing coronary artery bypass surgery. Success in this trial could establish direct mRNA delivery as a viable strategy to improve cardiac perfusion and function, although prior angiogenic gene therapies have highlighted the importance of optimizing dose, duration, and delivery.
Conclusion
The advancement of RNA therapeutics has been driven by high-throughput identification of target genes, chemical and structural modifications to enhance RNA stability and reduce toxicity, and improved delivery systems such as lipid nanoparticles. These innovations enable the development of therapies for previously undruggable targets, rapid vaccine generation, and personalized medicine.
Recognizing the translational potential, our hospital has established a fully integrated RNA Therapeutics program supporting both internal research and external academic or biotech collaborations. The program encompasses fundamental studies on mRNA sequence and structure, cGMP-compliant synthesis and purification, nanomedicine-based delivery systems, GLP preclinical testing, and clinical trial infrastructure spanning Phase 1 to Phase 3 studies. Partnerships with industry sponsors allow large-scale manufacturing for advanced clinical phases and commercialization. This comprehensive platform addresses a critical gap for small academic groups or startups lacking translational expertise, providing a seamless pathway from discovery to clinical application. Through this integrated approach, we aim to accelerate the development and clinical translation of innovative RNA-based therapies, realizing the full potential of RNA therapeutics.