the Delivery of mRNA-Based Vaccines
Vaccination has profoundly improved global health by reducing morbidity and mortality from infectious diseases. Traditional vaccine development follows the classical paradigm established by Louis Pasteur, often summarized as “isolate, inactivate, and inject” the causative pathogen [1]. Vaccines may be prophylactic or therapeutic and are generally categorized as live-attenuated (weakened microorganisms), inactivated (killed microorganisms), subunit (purified antigens), or toxoid (inactivated bacterial toxins). Modern vaccine strategies, such as subunit vaccines, aim to achieve comparable efficacy to conventional whole-cell vaccines while minimizing safety risks. However, subunit antigens typically exhibit lower immunogenicity, which can be enhanced using delivery systems or adjuvants to potentiate the immune response. Advances in genome-based rational vaccine design now provide significant opportunities to surpass the limitations of traditional whole-organism approaches.
The central dogma of molecular biology describes the flow of genetic information from DNA to mRNA and subsequently to protein . Gene therapy involves the delivery of genetic material into cells to correct defective genes, thereby curing or ameliorating disease . Vectors for gene therapy are broadly classified as viral or non-viral. Non-viral vectors offer advantages such as low immunogenicity and scalability , but their efficacy has historically been limited by challenges in plasmid DNA (pDNA) delivery, including nuclear entry, the presence of antibiotic resistance genes, and poor control over long-term expression. These limitations contrast with conventional drugs, which can be discontinued immediately, and pose risks of uncontrolled protein expression.
mRNA offers a solution to these challenges. It carries genetic information from the nucleus to the cytoplasm, where ribosomes translate it into protein. Unlike pDNA, mRNA is functional in both dividing and non-dividing cells and does not introduce extraneous genes, making it a safer vector. Its transient expression, typically lasting days to weeks depending on the platform, allows precise control over protein production . Additionally, cell-free mRNA manufacturing reduces contamination risks and facilitates production under good manufacturing practice conditions. mRNA therapeutics also avoid vector-induced immunogenicity seen with viral vectors or virus-like particles .
Current mRNA applications under investigation include
1. prophylactic and therapeutic vaccination against infectious diseases and cancer.
2. protein replacement therapy.
3. gene editing.
Two mRNA formats are commonly used: non-replicating mRNA, encoding only the antigen of interest, and self-amplifying mRNA, which additionally encodes replication machinery to enhance antigen expression . Self-amplifying mRNA platforms, often derived from single-stranded RNA viruses such as alphaviruses, flaviviruses, or picornaviruses, increase the magnitude and duration of protein expression and the resulting immune response . Non-replicating mRNA is primarily used for cancer vaccines, whereas both formats are applied in prophylactic vaccines for infectious diseases.
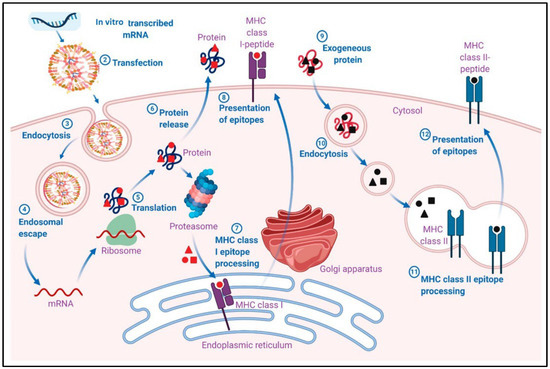
Therapeutically, in vitro transcribed (IVT) mRNA is used because it closely mimics fully mature cytosolic mRNA found in eukaryotic cells. IVT mRNA can be delivered either by ex vivo transfection of cells, which are subsequently adoptively transferred, or by direct in vivo administration to the cytosol. These strategies are being explored for genome editing, genetic reprogramming, adoptive T cell and dendritic cell (DC)-based immunotherapies for cancer and infectious diseases, tolerization protocols for allergy treatment, and protein replacement therapies. In both ex vivo and in vivo approaches, target cells synthesize the encoded protein(s) in situ, using the mRNA as a template. The coding sequence (open reading frame, ORF) of mature mRNA is flanked by untranslated regions (UTRs), and ideally includes a 5’ cap and a poly(A) tail.
The pharmacodynamic activity of both endogenous and IVT mRNA occurs in the cytosol. Unlike endogenous mRNA, which is transcribed in the nucleus and exported to the cytosol, IVT mRNA originates extracellularly and must enter the cytosol. Once delivered, IVT mRNA engages the same cellular machinery as native mRNA, enabling translation and post-translational modification of the encoded protein. For vaccines, these proteins function as antigens, eliciting pathogen-specific humoral and cell-mediated immune responses. The intracellular fate of the protein is directed by natural or engineered signal peptides or transmembrane domains, allowing mRNA vaccines to be designed for precise cellular localization and function.
The pharmacokinetics of IVT mRNA is influenced by both the mRNA half-life and the stability of the resulting protein. Two major challenges affecting cytosolic bioavailability are rapid RNase-mediated degradation and inefficient passive membrane diffusion due to molecular size and electrostatic repulsion from the negatively charged cell membrane. Naked mRNA is highly susceptible to extracellular RNases, compromising delivery and efficacy. Various transfection reagents and delivery systems, including cationic molecules, lipids, polymers, and nanoparticles, enhance cellular uptake, protect against degradation, and facilitate endosomal escape. Targeting DCs and physical methods such as electroporation have also improved delivery efficiency, particularly for large self-amplifying mRNA, as evidenced by enhanced reporter gene expression and immunogenicity in models using HIV envelope antigens. Natural nucleotide composition ensures IVT mRNA is metabolized by physiological pathways, minimizing metabolite-associated toxicity.
The development of mRNA-based therapeutics began in 1990 with the successful expression of various proteins following direct injection of mRNA into mouse muscle. This milestone led to the first mRNA-based vaccine tested in 1993, which induced an anti-influenza cytotoxic T-lymphocyte response in mice, and to the first vaccination using mRNA encoding cancer antigens in 1995. These early studies demonstrated the potential of mRNA for in situ protein expression and for eliciting protective, antigen-specific cellular and humoral immunity. However, the field remained largely dormant for nearly a decade until the capacity of in vivo mRNA administration to induce cytotoxic T lymphocytes and antibody responses was recognized. Progress was initially slow due to the labile nature of unmodified mRNA, which requires careful handling to avoid degradation, whereas DNA-based therapeutics were more stable and easier to work with.
In cell-free systems, mRNA is synthesized through in vitro transcription (IVT) of a DNA template, such as a linearized plasmid or PCR product, containing all the necessary structural elements of functional mRNA. IVT reactions require the DNA template, RNA polymerase, and nucleotide building blocks. Following transcription, the DNA template is typically removed using DNases, and the mRNA is purified using methods such as precipitation or chromatography, yielding highly pure mRNA suitable for therapeutic use. Multiple strategies have since been developed to address the intrinsic instability and potential immunogenicity of mRNA, including chemical modifications and formulation techniques to enhance stability and translation efficiency.
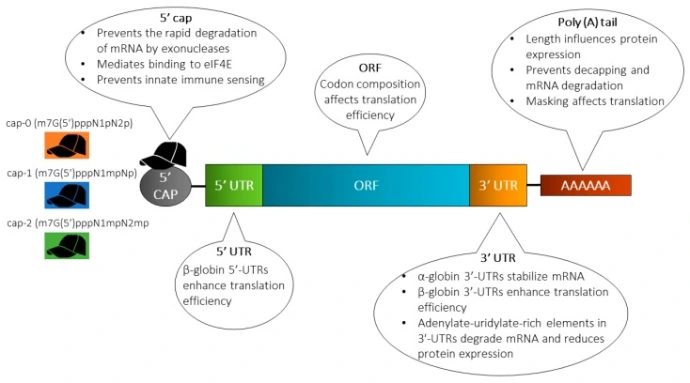
Molecular Stabilization
Strategies to enhance mRNA stability and translation are critical for increasing protein expression. These approaches include elongating the poly(A) tail, modifying the 5’ cap, engineering the untranslated regions (UTRs) and coding sequence (ORF), and incorporating chemically modified nucleotides. Such sequence and structural optimizations extend mRNA half-life and improve translational efficiency.
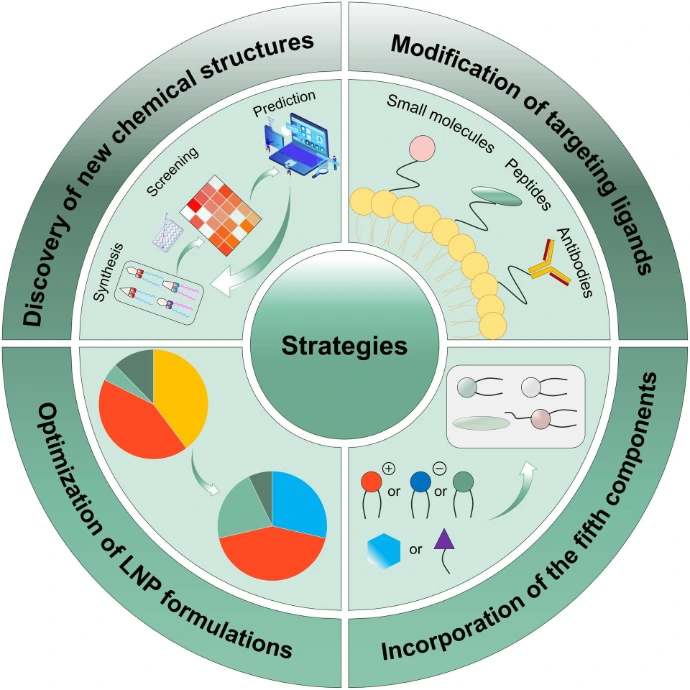
Formulation Strategies
Efficient intracellular delivery of mRNA remains a major challenge, particularly for systemic administration. The large molecular weight and high negative charge of mRNA hinder its passage across cellular membranes. Without a delivery system, mRNA absorption is extremely low, and its half-life is short. Additionally, mRNA is inherently unstable and highly susceptible to degradation by 5’ exonucleases, 3’ exonucleases, and endonucleases. Therefore, delivery systems are essential for transporting mRNA to the cytosol both in vitro and in vivo. Various strategies have been explored to improve RNA delivery, including microinjections, RNA patches, gene gun administration, protamine condensation, RNA adjuvants, and encapsulation within lipid or polymer-based nanoparticles. Typically, IVT mRNA is formulated with a delivery system by complexing it with protective agents that prevent rapid degradation and promote cellular uptake. While efficient carriers are generally considered necessary for effective in vivo transfection, naked mRNA has also been successfully applied in some studies. The following sections will first address the delivery of naked mRNA, followed by discussions on vector-based mRNA delivery strategies.
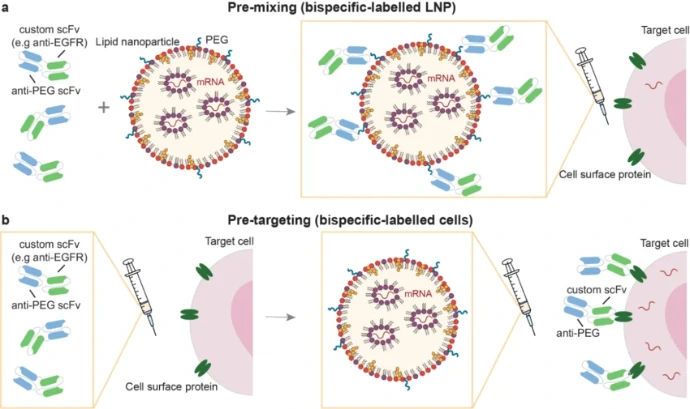
Cell-Specific mRNA Delivery
Targeted delivery of mRNA to specific cell types enhances therapeutic efficacy by increasing uptake in the desired cells, reducing the required dose, and minimizing off-target effects. Lymphoid organs can be selectively targeted by modulating the net charge of the formulation, exploiting the proximity of antigen-presenting cells (APCs) to T cells to optimize priming and amplification of T-cell responses. Active targeting of mRNA-loaded nanoparticles has been shown to induce robust effector and memory T-cell responses, including IFN-α-mediated rejection of tumors. In one approach, mRNA encoding FLuc or IL-10 was selectively delivered to Ly6c+ leukocytes by coating lipid nanoparticles (LNPs) with anti-Ly6c monoclonal antibodies. Alternatively, dendritic cells and macrophages can be targeted via C-type lectin receptors, which recognize mannose- or fucose-terminated glycans and mediate endocytosis. This principle has been applied to transfect GFP mRNA into dendritic cells using mannose-cholesterol conjugates assembled with polyethylene glycol (PEG) linkers.
Conclusion
The field of mRNA-based therapeutics encompasses applications ranging from protein replacement and gene editing to prophylactic and therapeutic vaccination. With numerous mRNA vaccine candidates currently in preclinical and clinical development, mRNA technology has demonstrated significant potential for generating novel vaccines against infectious diseases and cancer. Nonetheless, challenges related to mRNA’s large molecular size, negative charge, intrinsic instability, and high susceptibility to enzymatic degradation continue to limit its clinical translation. Effective application of mRNA therapeutics depends on improved delivery systems capable of enhancing stability, cellular targeting, and translational efficiency. While advanced formulations such as lipoplexes, lipid nanoparticles, polymers, and lipid-polymer hybrids have shown promise in terms of safety, stability, transfection efficiency, and cost, many clinical candidates still rely on naked mRNA delivery, highlighting the need for further optimization. Continued innovation in mRNA formulation and nanomaterial-based delivery strategies is expected to expand the therapeutic and prophylactic potential of mRNA for the treatment and prevention of infectious diseases and cancers.