Antisense Oligonucleotides
Many diseases arise from abnormal protein production or metabolism. Since proteins are synthesized from messenger RNA (mRNA), regulating mRNA provides a direct route to control aberrant protein expression. Beyond mRNA, non-coding RNAs (ncRNAs) including microRNAs (miRNAs), transfer RNA-derived small RNAs, PIWI-interacting RNAs (piRNAs), long ncRNAs (lncRNAs), circular RNAs, and pseudogenes play critical roles in controlling gene expression and cellular function.
Antisense strategies target pre-mRNA, mRNA, or ncRNAs to modulate disease causing proteins. Unlike traditional small-molecule drugs that bind proteins, antisense therapies use Watson–Crick base pairing to recognize RNA sequences, offering greater precision and flexibility in drug design. Chemical modifications of oligonucleotides, combined with innovative delivery methods, have further enhanced the effectiveness of these therapies.
With several nucleic acid-based drugs already approved by the FDA, antisense research has gained significant momentum. Today, numerous antisense candidates are in clinical trials for cardiovascular, metabolic, neurological, neuromuscular, inflammatory, and infectious diseases. This overview highlights the structural innovations, mechanisms, delivery strategies, FDA-approved therapies, and emerging antisense drugs in development.
Early antisense oligonucleotides (ASOs) with a natural phosphodiester backbone were able to target RNA, but only with moderate effectiveness. Their phosphodiester bonds made them highly susceptible to degradation by nucleases, and their size and negative charge limited cellular uptake. To overcome these challenges, newer generations of chemically modified ASOs have been developed. These modifications enhance stability, improve therapeutic efficacy, and reduce immune activation and off-target effects.
1. Phosphorothioate (PS) Modifications
Phosphorothioate (PS) ASOs are first-generation antisense oligonucleotides that function primarily through mRNA cleavage. In PS-ASOs, one of the non-bridging oxygen atoms in the phosphate backbone is replaced by sulfur, making the molecule more resistant to nuclease degradation. Compared to unmodified ASOs, PS-ASOs bind strongly to serum proteins like albumin, reducing renal clearance and extending circulation time in the body.
Pharmacokinetic studies show that after intravenous administration in mice, about 40% of PS-ASOs are excreted in urine within 48 hours, and systemic delivery leads to higher accumulation in organs such as the liver, kidney, and spleen, with efficient cellular uptake. In monkeys, PS-ASOs exhibit biphasic plasma elimination with an initial half-life of 30–48 minutes, followed by a longer half-life of 35–50 hours. However, repeated administration in rodents can trigger immune responses, indicating potential toxicity concerns.
Phosphorodiamidate morpholino oligomers (PMOs) are antisense oligonucleotides in which the usual five-membered sugar is replaced with a six-membered morpholine ring, connected through phosphorodiamidate linkages. PMOs are charge-neutral and act mainly by steric blocking or modulating RNA splicing. Their unique structure, lacking a carbonyl group, makes them resistant to proteases and esterases, while the morpholine ring improves water solubility. PMOs have been widely used in developmental biology and other antisense-based applications due to their stability and versatility.
Peptide nucleic acids (PNAs) are synthetic analogs of nucleic acids in which the sugar-phosphate backbone is replaced by neutral N-2-aminoethyl glycine units, with nucleobases linked via flexible methyl carbonyl bridges. This unique, unnatural backbone makes PNAs resistant to enzymatic degradation and gives them a stronger binding affinity for RNA compared to unmodified ASOs. However, PNAs face challenges such as low water solubility and poor cellular uptake. To overcome these limitations, researchers have developed chemically modified PNAs, including cationic PNAs, alpha- and gamma-guanidinium PNAs, and lysine-conjugated PNAs, improving both solubility and cell penetration.
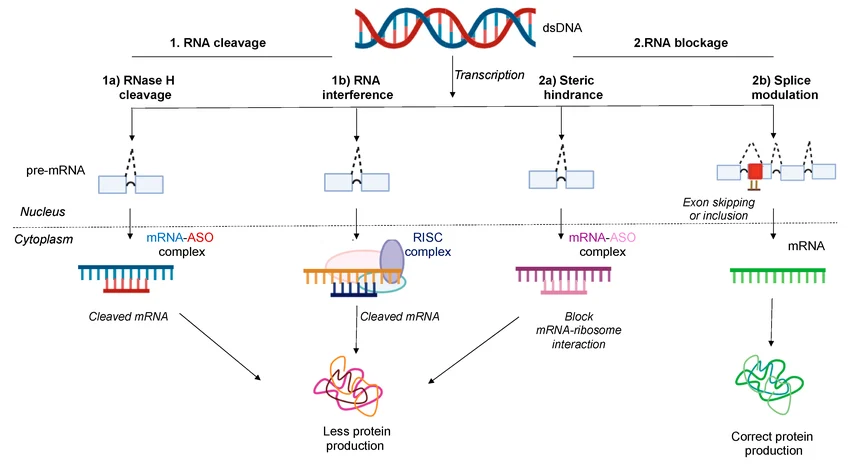
Nearly 40 years ago, Zamecnik and Stephenson demonstrated that short, synthetic single-stranded ASOs could block protein production by targeting viral mRNA. Today, it is well established that active ASOs are typically 15–20 nucleotides long and selectively bind complementary RNA sequences via Watson–Crick base pairing, minimizing off-target effects. ASOs exert their therapeutic effects through two main mechanisms:
1- RNA cleavage, where the target RNA is degraded.
2- RNA blockage, where RNA function is sterically inhibited without degradation.
MicroRNAs (miRNAs) are 20–25 nucleotide-long noncoding RNAs that regulate cell identity and function. Dysregulation of miRNAs is linked to various diseases, both malignant and non-malignant. Antisense oligonucleotides (ASOs) have been designed to target specific miRNAs for therapeutic purposes.
For example, targets miR-122 to treat hepatitis C virus (HCV) infection using LNA and PS chemistry, though severe side effects halted its clinical trials. RG-012 (Regulus Therapeutics) targets miR-21 to slow renal fibrosis in Alport syndrome and has orphan drug status in the US and Europe.
In oncology, miRNAs that drive cancer progression are targeted as well. Cobomarsen (MRG-106), an LNA-based inhibitor of miR-155 developed by miRagen Therapeutics, is in phase II trials for cutaneous T-cell lymphoma, adult T-cell lymphoma, and leukemia.
These examples highlight how ASOs can modulate miRNA activity to treat viral infections, kidney diseases, and cancers.
Antisense oligonucleotides (ASOs) have become a powerful type of therapy. Early challenges with low effectiveness and the need for high doses have been overcome through advances in chemistry and biology, improving stability, strength, and reducing side effects. These improvements have led to multiple FDA and EMA-approved ASO drugs, including treatments for rare and genetic disorders like hATTR (Patisiran, Inotersen) and Duchenne muscular dystrophy (Golodirsen, Eteplirsen).
ASOs have unique advantages: flexible design using base pairing, precise targeting of disease-causing RNA, and potential for both personalized treatments (like Milasen) and therapies for larger patient groups (like Inclisiran). Unlike traditional drugs that require daily dosing, ASOs can be given weekly or even once every few months, making treatment easier for patients.
Challenges remain, especially in delivery. ASOs often accumulate in organs like the liver, kidney, and spleen, which can cause side effects. New delivery methods are important to guide ASOs to the right tissues, reduce doses, and improve effectiveness. Manufacturing at large scale also needs to be consistent and reliable.
ASOs are also being explored for antiviral therapies, targeting multiple genes of a virus or helping the body’s immune response to fight infection.
In short, ASOs are gaining importance in medicine, and with better delivery systems, they have the potential to safely treat both rare and widespread diseases.