RNA-Based Therapies in Cancer Immunotherapy
Overview of Modern Cancer Immunotherapy
In recent years, cancer immunotherapy has emerged as one of the most transformative approaches in oncology. Unlike conventional treatments such as chemotherapy and radiotherapy which directly target tumor cells but often damage healthy tissues immunotherapy harnesses the body’s own immune system to identify, attack, and eliminate malignant cells.
Major breakthroughs have accelerated the clinical adoption of immunotherapy. The approval of immune checkpoint inhibitors such as pembrolizumab (2014) and nivolumab (2015), along with CAR-T cell therapies like tisagenlecleucel (2017) and axicabtagene ciloleucel (2018), has demonstrated the remarkable potential of immune-based strategies in treating cancer.
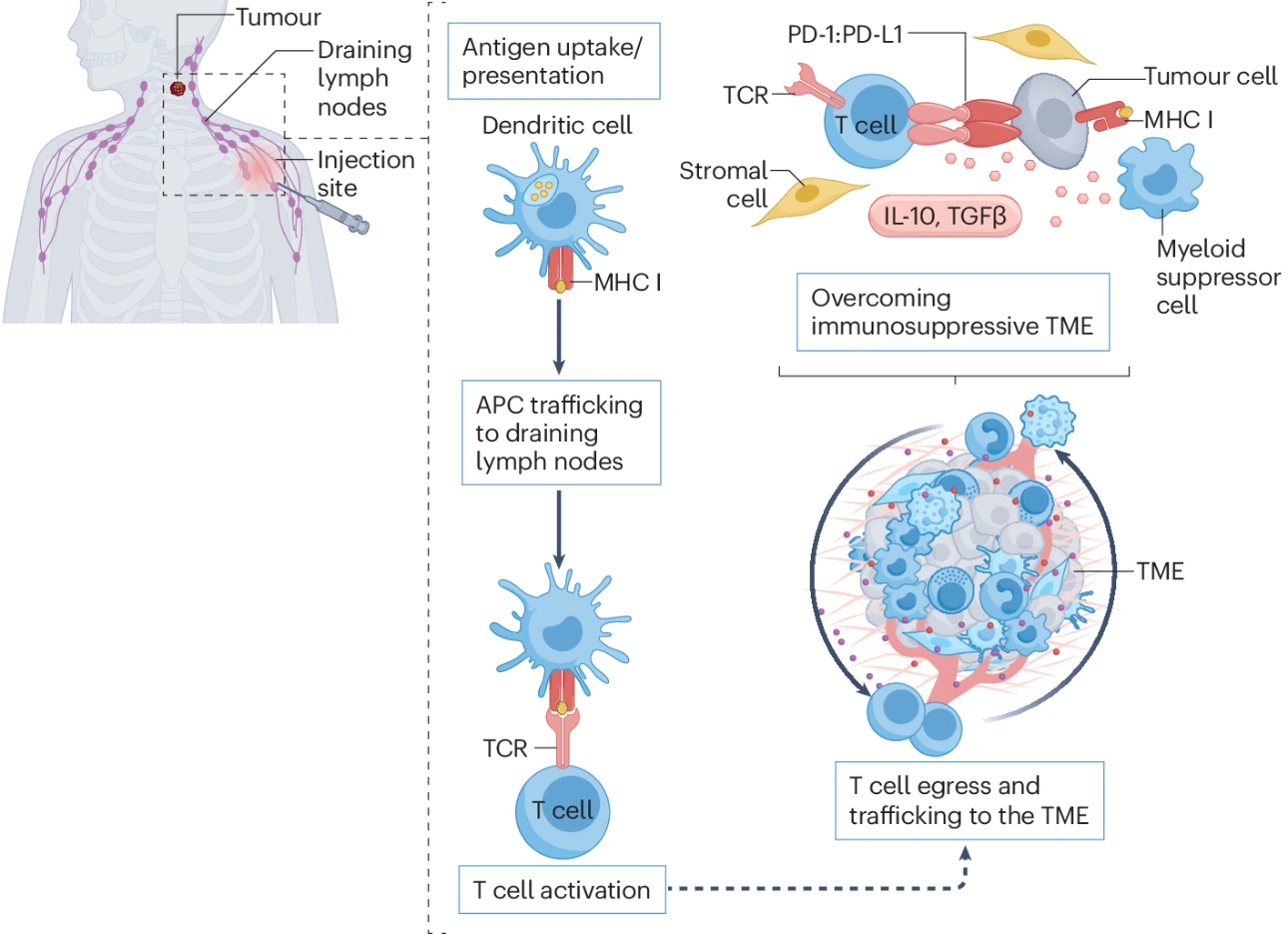
These therapies work by enhancing immune recognition and overcoming tumor-induced immunosuppression, enabling more effective and durable anti-tumor responses.
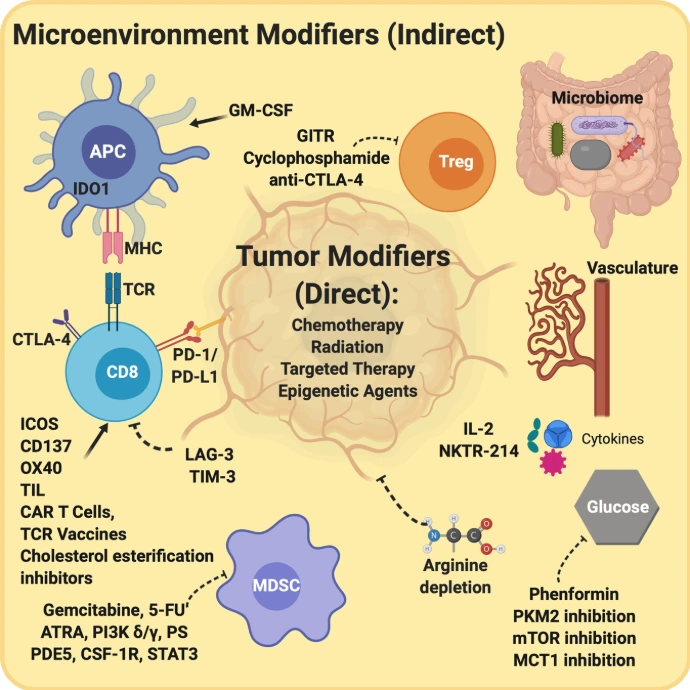
Therapeutic Goals of Cancer Immunotherapy
The primary objective of cancer immunotherapy is to:
- Strengthen the host’s anti-tumor immune response
- Reprogram the tumor microenvironment from immunosuppressive to immunoreactive
- Suppress tumor growth and metastasis
- Improve long-term survival and quality of life
Unlike traditional treatments, immunotherapies can induce long-lasting immune memory, allowing the immune system to continue recognizing cancer cells even after treatment ends. This opens the possibility of sustained remission and, in some cases, functional cures.
Cancer Vaccines
Cancer vaccines represent a highly promising branch of immunotherapy. Unlike conventional vaccines that prevent infectious diseases, cancer vaccines are designed to:
- Train the immune system to recognize tumor-specific markers
- Prevent cancer development in high-risk individuals
- Treat existing malignancies by stimulating targeted immune responses
Targeting Tumor Antigens
Cancer vaccines often focus on:
- Tumor-Associated Antigens (TAAs)
- Tumor-Specific Antigens (TSAs)
These molecules are uniquely expressed or overexpressed in cancer cells, making them ideal therapeutic targets. By introducing these antigens into the body, vaccines “educate” immune cells especially T lymphocytes to selectively attack tumor cells while sparing healthy tissues.
Role of Immunological Memory in Vaccine Efficacy
A critical advantage of cancer vaccines is their ability to induce immunological memory. This means:
- The immune system retains long-term recognition of tumor antigens
- Rapid and robust responses occur upon cancer recurrence
- Reduced need for repeated treatments
Additionally, cancer vaccines are generally well tolerated, with fewer systemic toxicities compared to chemotherapy.
Challenges in Cancer Vaccine Development
Despite decades of research, translating cancer vaccines into widely effective clinical therapies remains challenging due to:
- High variability of tumor antigens between patients
- Tumor immune evasion mechanisms
- Weak or insufficient immune activation in early designs
However, advances in genomics, bioinformatics, and molecular biology are enabling more precise antigen identification and improved vaccine formulations.
Approved and Emerging Cancer Vaccines
Preventive vaccines targeting oncogenic viruses such as those against human papillomavirus (HPV) and hepatitis B virus (HBV) have demonstrated success in reducing cancer incidence.
In the therapeutic space, sipuleucel-T (PROVENGE) became the first FDA-approved cancer vaccine for prostate cancer, marking a milestone in personalized immunotherapy.
Ongoing clinical trials are now exploring:
- Personalized neoantigen vaccines
- Combination therapies with checkpoint inhibitors
- Cytokine-enhanced vaccine platforms
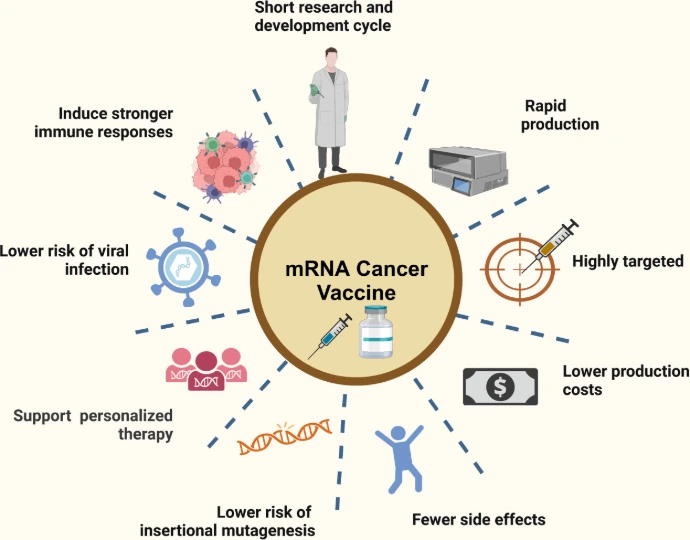
Why RNA-Based Vaccines Are Revolutionary
RNA-based vaccines offer several unique advantages over traditional platforms:
mRNA vaccines can encode multiple tumor antigens simultaneously, increasing the likelihood of overcoming tumor heterogeneity and resistance.
They stimulate both:
- Humoral immunity (antibody-mediated)
- Cellular immunity (T-cell mediated)
Unlike peptide vaccines, mRNA vaccines are less restricted by human leukocyte antigen (HLA) variability, making them applicable across diverse patient populations.
Clinical Progress and Future Potential
More than 20 mRNA-based cancer immunotherapies are currently in clinical trials, targeting both solid tumors and hematological malignancies. Early results indicate:
- Strong immune activation
- Favorable safety profiles
- Potential synergy with existing immunotherapies
The global success of mRNA vaccines during the pandemic has further accelerated interest in applying this technology to cancer treatment
Conclusion
RNA-based therapeutics represent a major shift toward precision oncology. Their flexibility, scalability, and ability to encode personalized tumor antigens position them as a cornerstone of next-generation cancer treatments.
While challenges remain particularly in delivery optimization and immune modulation ongoing research continues to refine these platforms. The integration of RNA vaccines with other immunotherapies, such as checkpoint inhibitors and adoptive cell transfer, is expected to unlock even greater therapeutic potential.
Ultimately, RNA-based cancer immunotherapy holds promise not only for improving survival outcomes but also for redefining how cancer is treated moving from generalized therapies to highly personalized, immune-driven solutions.