RNAi-Based Therapeutics
MicroRNAs (miRNAs) and small interfering RNAs (siRNAs) are natural regulators of gene expression that act at the posttranscriptional level. RNA interference (RNAi) technology harnesses these molecules to precisely modulate disease-related genes, offering new opportunities for targeted therapies. Four siRNA drugs—patisiran, givosiran, lumasiran, and inclisiran are FDA-approved, and many others are in clinical trials.
Despite their potential, RNA therapeutics face delivery and stability challenges in the body. Advances in chemical modifications, delivery systems, and in vivo production of authentic RNA molecules are helping overcome these barriers, enabling safer, more effective treatments. These innovations position RNAi as a versatile platform for research, precision medicine, and novel therapeutic development.
Genome-derived microRNAs (miRNAs) and exogenous small interfering RNAs (siRNAs) are key noncoding RNA molecules that regulate gene expression at the posttranscriptional level. miRNAs, typically 18–25 nucleotides long, can target multiple mRNA transcripts to modulate several cellular pathways simultaneously. In contrast, exogenous siRNAs are usually designed to selectively silence a single gene. These RNAi molecules play essential roles in controlling diverse cellular processes, including signaling, proliferation, metabolism, immune responses, and senescence.
Genome-derived microRNAs (miRNAs) are small noncoding RNAs that regulate gene expression posttranscriptionally. Their biogenesis occurs in two main phases: nuclear and cytosolic. miRNAs can originate from intergenic regions, with their own promoters, or from intragenic regions within protein-coding genes, coexpressed with their host genes. In the nucleus, miRNA genes are transcribed into primary miRNAs (pri-miRNAs), which are processed by Drosha and DGCR8 into precursor miRNAs (pre-miRNAs). Pre-miRNAs are exported to the cytoplasm by Exportin-5 and further cleaved by Dicer/TRBP into mature miRNA duplexes. The guide strand is loaded into the RNA-induced silencing complex (RISC), while the passenger strand is degraded, enabling sequence-specific regulation of target mRNAs.
In addition to this canonical pathway, miRNAs can also arise via noncanonical mechanisms that bypass Drosha/DGCR8 or Dicer/TRBP processing, such as mirtrons, or derive from other RNA species like small nucleolar RNAs (snoRNAs) and transfer RNAs (tRNAs). tRNA-derived fragments (tRFs) and snoRNA-derived miRNAs can also associate with RISC to regulate gene expression, demonstrating the diversity and flexibility of miRNA-mediated gene regulation.
Exogenous small interfering RNAs (siRNAs) are synthetic, gene-specific RNA molecules that regulate target mRNA expression through the RNA interference (RNAi) pathway. Typically 20–25 nucleotides in length with 3′ overhangs, siRNAs can bypass Dicer processing and directly incorporate into the RNA-induced silencing complex (RISC) to suppress a specific gene, often one considered “undruggable” by conventional small molecules.
siRNAs can be produced through chemical synthesis, such as solid-phase synthesis, which allows precise sequence design and incorporation of chemical modifications, or through enzymatic in vitro transcription (IVT) from DNA templates, followed by duplex formation and Dicer processing. Chemical modifications enhance siRNA stability, reduce immune activation, improve potency, and enable tissue-specific targeting. Common modifications include alterations to the ribose ( 2′-O-methyl, 2′-fluoro), nucleobases, or the phosphodiester backbone, which improve nuclease resistance, cellular uptake, and endosomal escape.
Modern synthetic siRNAs leverage these strategies to create highly stable, efficient, and safe molecules suitable for research and clinical applications. These advances underpin several FDA-approved siRNA therapeutics and numerous candidates in clinical development.
Both microRNAs (miRNAs) and small interfering RNAs (siRNAs) regulate gene expression at the posttranscriptional level through the RNA-induced silencing complex (RISC). The type of regulation depends on the RNA agent: siRNAs typically target a single specific mRNA for cleavage and degradation, while miRNAs can target multiple mRNAs, leading to translational repression, mRNA degradation, or occasionally activation.
miRNAs generally bind imperfectly to the 3′ untranslated region (3′UTR) of target mRNAs, allowing broad regulation of multiple genes. In contrast, siRNAs usually bind perfectly to the coding region of a single mRNA, ensuring precise gene silencing. Notably, some FDA-approved siRNA therapeutics mimic miRNA-like targeting by binding to the 3′UTR with perfect complementarity to induce transcript cleavage.
The discovery of microRNAs as key regulators of gene expression has opened new avenues for oligonucleotide based therapies. Unlike siRNAs, which typically target a single gene, miRNA-targeted therapies can modulate entire cellular pathways, offering broader therapeutic potential.
Therapeutic strategies include:
miRNA replacement: Supplementing downregulated or non-functional miRNAs with synthetic oligonucleotides to restore normal cellular function.
miRNA inhibition: Using artificial antagonists either oligonucleotides or small molecules to counteract the effects of overexpressed, disease promoting miRNAs.
For effective clinical application, chemical modifications of oligonucleotides, combined with efficient delivery systems, are essential. While miRNA therapies leverage decades of research from other oligonucleotide therapeutics, challenges remain, particularly due to the short sequences of miRNA targets, which can complicate specificity and stability.
Current developments highlight the potential of miRNA-targeted therapies in treating cancers, viral infections, and other complex diseases, with ongoing research focused on improving delivery, safety, and efficacy.
The link between microRNAs (miRNAs) and numerous human diseases, particularly cancers, has sparked growing interest in their potential as novel therapeutics. This has driven interdisciplinary research in biology, chemistry, and medicine to develop miRNA-based treatments. The recent success of mRNA vaccines has also revitalized interest in RNA-based immunotherapies, positioning miRNAs as adaptable and modular options for cancer therapy.
Advances in RNA chemistry and delivery technologies are central to shaping miRNA therapeutics. Despite promising preclinical results, miRNA-based therapies are still in early development. Few have progressed to clinical trials, none have reached Phase III, and some have been halted due to toxicity concerns.
Key challenges remain:
Ensuring target specificity and selectivity
Minimizing immunogenicity and off-target effects
Developing efficient delivery systems
Determining safe and effective dosing
Expanding understanding of miRNA functions for clinical use
Addressing these challenges is essential for translating the therapeutic potential of miRNAs into safe and effective treatments for cancer and other diseases.

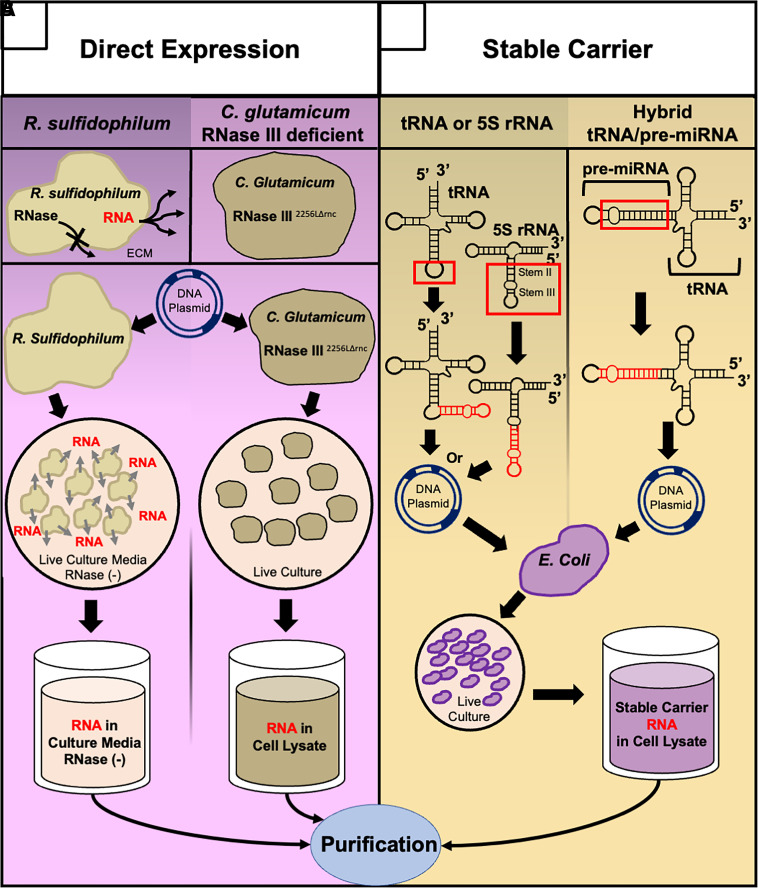
Traditional chemical and biochemical synthesis methods for RNA molecules remain widely used, but they may alter the natural properties of RNAi agents, potentially affecting their efficacy and safety. To overcome this, new biotechnologies aim to produce RNAi molecules within living cells, generating more biologically authentic RNA.
Two main approaches are emerging:
Direct expression in host cells: using engineered bacteria or other host strains to produce RNA naturally.
RNA production via stable carriers :leveraging cellular systems or carriers to generate RNAi agents with structures and modifications closely resembling their natural counterparts.
These innovations promise safer, more effective RNAi therapeutics that better mimic endogenous RNA function.
RNAi has proven to be a highly adaptable tool for gene regulation, reverse genetics, and the development of targeted therapies. The RNAi pathway is not static but consists of versatile components that can be engineered to improve potency, stability, and therapeutic function. Recent FDA approvals of siRNA drugs, such as inclisiran, and the growing number of RNAi therapeutics in clinical trials highlight its clinical potential.
Despite this progress, miRNA-based therapies remain underexplored. Unlike siRNAs, miRNAs can target multiple mRNAs, offering the potential to modulate entire biological pathways with a single therapeutic. Achieving this requires careful research to optimize dosing, specificity, and safety.
Innovative production methods, including in vivo RNA expression in host cells and stable RNA carriers, are advancing the field by preserving natural RNA properties while maintaining stability and activity. Platforms such as tRNA/pre-miRNA carriers allow the generation of biologically active miRNAs, siRNAs, and other RNA molecules, better mimicking natural RNA for therapeutic use.
Future research will determine the clinical efficacy of these bioengineered RNAi molecules, potentially enabling new therapies for previously “undruggable” targets and expanding the reach of RNA-based medicine.