Intracellular RNA delivery by lipid nanoparticles
Introduction
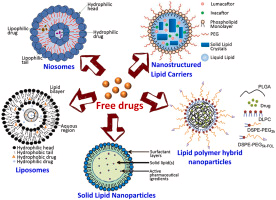
Lipid nanoparticles (LNPs) and other nanocarriers are promising tools for delivering RNA molecules such as siRNA, miRNA, and mRNA into cells. Their lipid-based structure enables membrane compatibility, stability, and tunable properties for efficient cellular uptake and RNA release. LNPs may have multilamellar, cubic, or inverse hexagonal structures, with RNA interacting mainly with hydrophilic lipid heads. RNA release occurs via diffusion or particle degradation, but the detailed mechanisms are still being studied. Phenomenological models help describe these processes and guide the design of more effective RNA delivery systems.
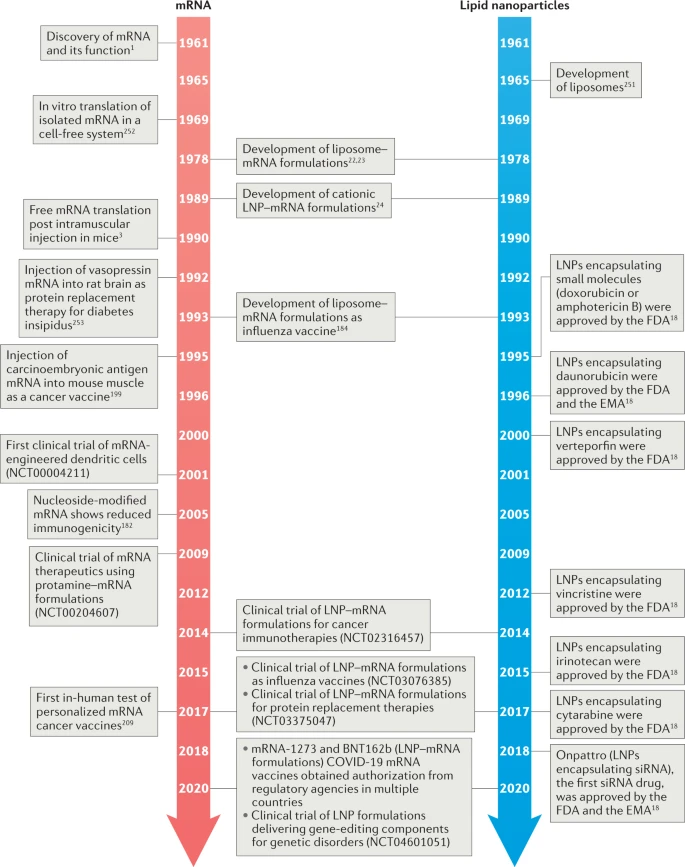
Messenger RNA (mRNA) plays a key role in biology by acting as the temporary link between genetic information and protein production. First identified through pioneering research between 1947 and 1961, mRNA carries the instructions from genes that cells use to build proteins.
In the late 1980s, deeper research into mRNA structure and function led to the creation of in vitro–transcribed (IVT) mRNA, a laboratory-produced version that can be used for therapeutic purposes. Since the first successful proof-of-concept experiments in animals in 1990, scientists have developed many approaches to improve the stability and safety of IVT mRNA, particularly by reducing its tendency to degrade quickly or trigger unwanted immune responses.
At the same time, major advances in drug delivery technologies have accelerated the development of mRNA-based treatments. Together, these innovations have positioned mRNA as a promising new class of therapeutic molecules with potential applications in vaccines, genetic diseases, and many other medical fields.
Lipid-Based Nanocarriers and Their Biomedical Applications
Lipid vesicles, commonly referred to as liposomes, play a critical role in both fundamental research and applied biomedical technologies. One important application is the formation of supported lipid bilayers, which serve as experimental models for studying membrane dynamics, protein interactions, and drug screening processes.
In addition to their structural utility, liposomes can exhibit intrinsic antimicrobial properties. These properties can be further enhanced when liposomes are used as carriers for encapsulated drugs, improving drug stability, bioavailability, and therapeutic efficacy.
More broadly, lipid-based nanocarriers have emerged as a cornerstone of next-generation drug delivery systems. Their ability to encapsulate and protect sensitive biomolecules enables the targeted delivery of therapeutics that would otherwise be rapidly degraded in biological environments.
RNA Diffusion from Nanopores
RNA molecules used in medicine can have very different sizes. Small RNAs, such as siRNA and miRNA, contain around 20–25 nucleotides, while mRNA molecules are much longer and can reach up to about 1000 nucleotides. Because of their small size, siRNA and miRNA are usually almost linear in solution, whereas mRNA tends to fold into more complex shapes.
Inside lipid nanoparticles (LNPs), these RNA molecules can be stored in tiny channels called nanopores. Small RNAs easily fit into these narrow spaces, while larger mRNAs may need to partially unfold to fit inside. This unfolding can happen because of the limited space inside the nanopores and the attraction between negatively charged RNA and positively charged lipids in the nanoparticle.
For RNA therapies to work, the RNA must leave the nanoparticle after it enters the cell. This release process can occur through diffusion, where the RNA slowly moves out of the nanopores. However, the speed of this process depends on how strongly the RNA interacts with the nanoparticle walls.
Research suggests that small RNAs can be released relatively easily, as long as the interaction between RNA and the nanoparticle wall is weak. In contrast, mRNA release is more complex, because stronger interactions may prevent it from escaping efficiently. For this reason, additional mechanisms such as nanoparticle degradation or special responsive materials may be needed to ensure effective mRNA delivery inside cells.
RNA Diffusion and Lipid Nanoparticle (LNP) Degradation
Lipid nanoparticles (LNPs) are currently the most widely used and clinically advanced system for delivering RNA molecules. Although many studies have focused on improving RNA delivery to enhance protein production, the relationship between biological stability, targeting accuracy, and therapeutic effectiveness of RNA-LNP systems is still not fully understood.
This overview emphasizes the important role of LNP stability in biological environments, particularly when nanoparticles interact with bodily fluids. Such interactions can strongly influence how efficiently RNA is delivered to cells. Key factors affecting LNP behavior include lipid composition, surface characteristics of the particles, and interactions with proteins present in physiological conditions.
Different analytical techniques are used to study the stability of RNA-LNPs in biological fluids, including dynamic light scattering, liquid chromatography, and fluorescence or radiolabel-based methods.
Finally, researchers are exploring strategies to improve nanoparticle stability, such as protective lipid coatings and shielding lipids, which can help maintain particle integrity during circulation in the body. Understanding the balance between LNP stability and controlled instability is essential for designing more effective RNA delivery systems and improving their performance in real biological conditions.

Conclusion
Lipid nanoparticles represent one of the most advanced and promising platforms for intracellular RNA delivery. Their unique combination of biocompatibility, structural versatility, and tunability makes them indispensable in modern therapeutic strategies, particularly in the rapidly evolving field of RNA medicine.
However, critical challenges remain especially in understanding and controlling RNA release mechanisms within cells. Continued integration of experimental studies with theoretical modeling will be essential to fully unlock the potential of LNP-based delivery systems and to develop more effective, targeted, and safe RNA therapies in the future.